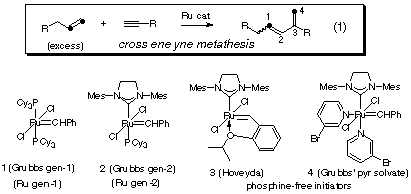
Ring closing enyne metathesis : A powerful tool for the synthesis of heterocycles - Chemical Society Reviews (RSC Publishing) DOI:10.1039/B508899M
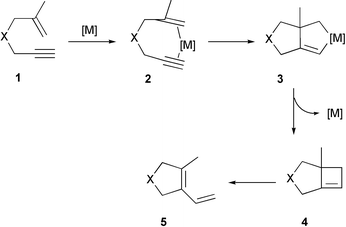
Ring closing enyne metathesis : A powerful tool for the synthesis of heterocycles - Chemical Society Reviews (RSC Publishing) DOI:10.1039/B508899M
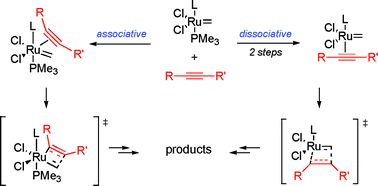
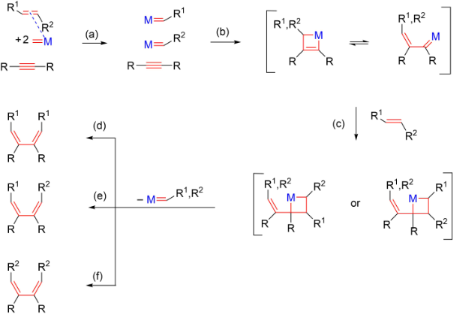
Feasibility of associative mechanism in enyne metathesis catalyzed by grubbs complexes - Dalton Transactions (RSC Publishing)
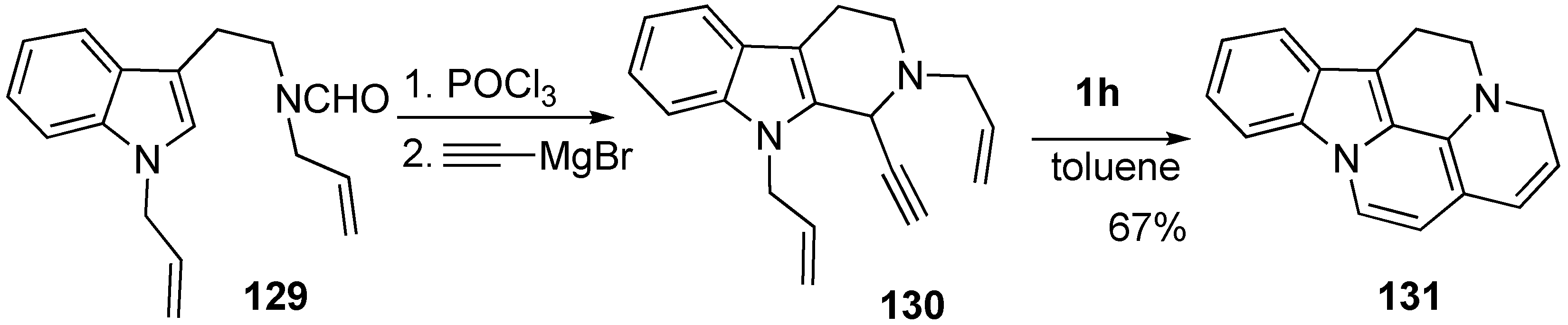
Materials | Free Full-Text | Recent Progress on Enyne Metathesis: Its Application to Syntheses of Natural Products and Related Compounds | HTML
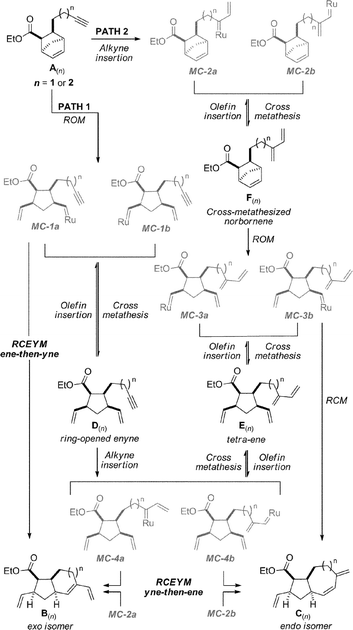
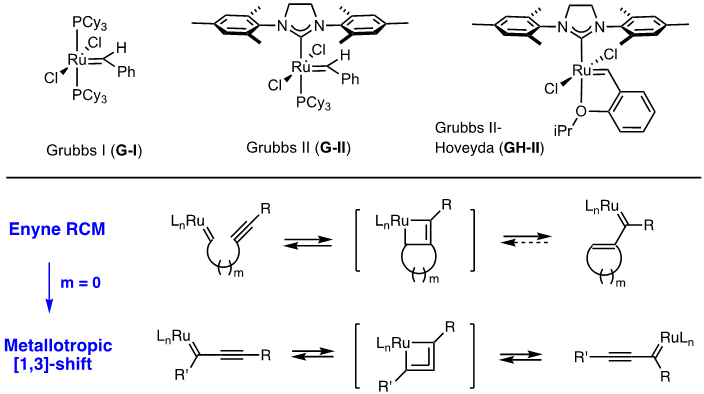
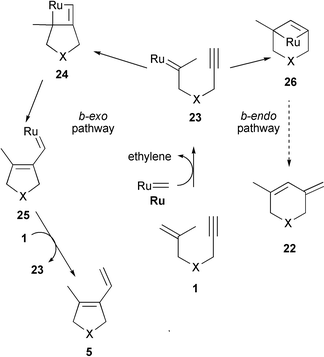
Exploiting domino enyne metathesis mechanisms for skeletal diversity generation - Chemical Communications (RSC Publishing) DOI:10.1039/B807278G

Modular Approach to Degradable Acetal Polymers Using Cascade Enyne Metathesis Polymerization - Fu - 2019 - Angewandte Chemie International Edition - Wiley Online Library
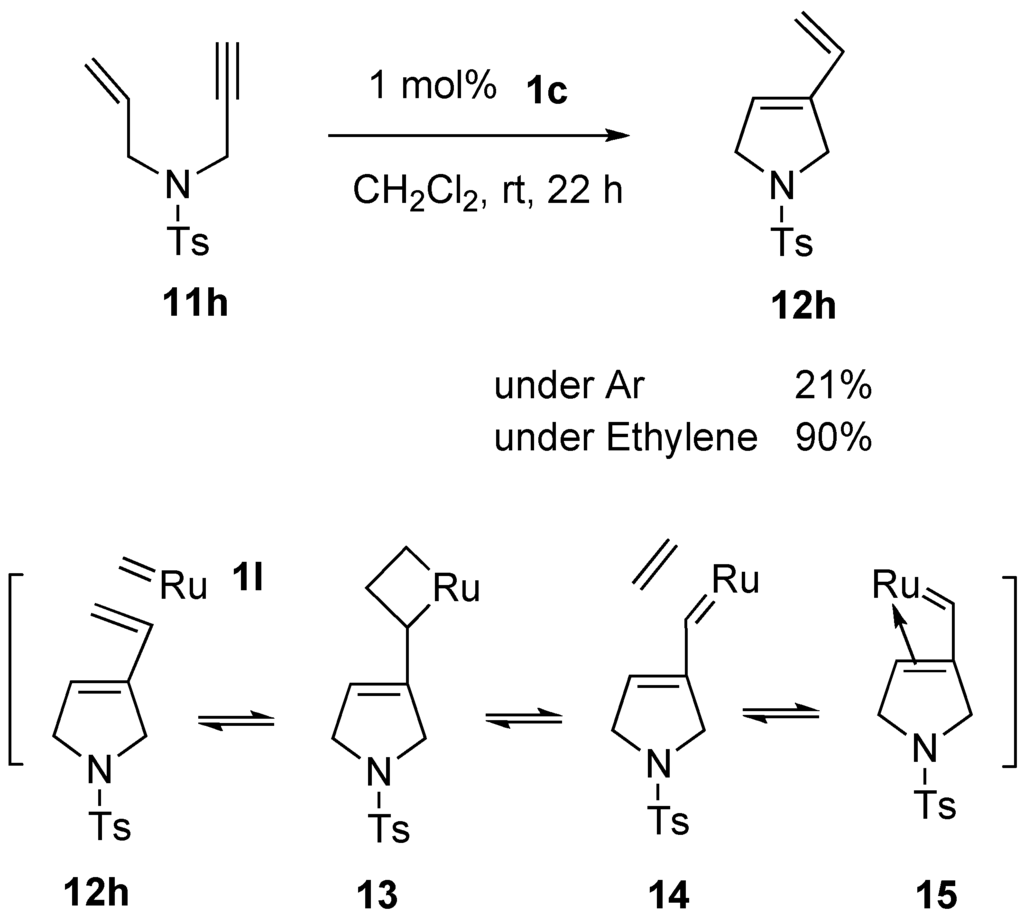
Materials | Free Full-Text | Recent Progress on Enyne Metathesis: Its Application to Syntheses of Natural Products and Related Compounds | HTML

Ring‐Closing Metathesis Dimerizations of Enynes and Deprotections of Propargyl Ethers Mediated by Carbene Ruthenium Complexes - Hahn - 2005 - European Journal of Organic Chemistry - Wiley Online Library