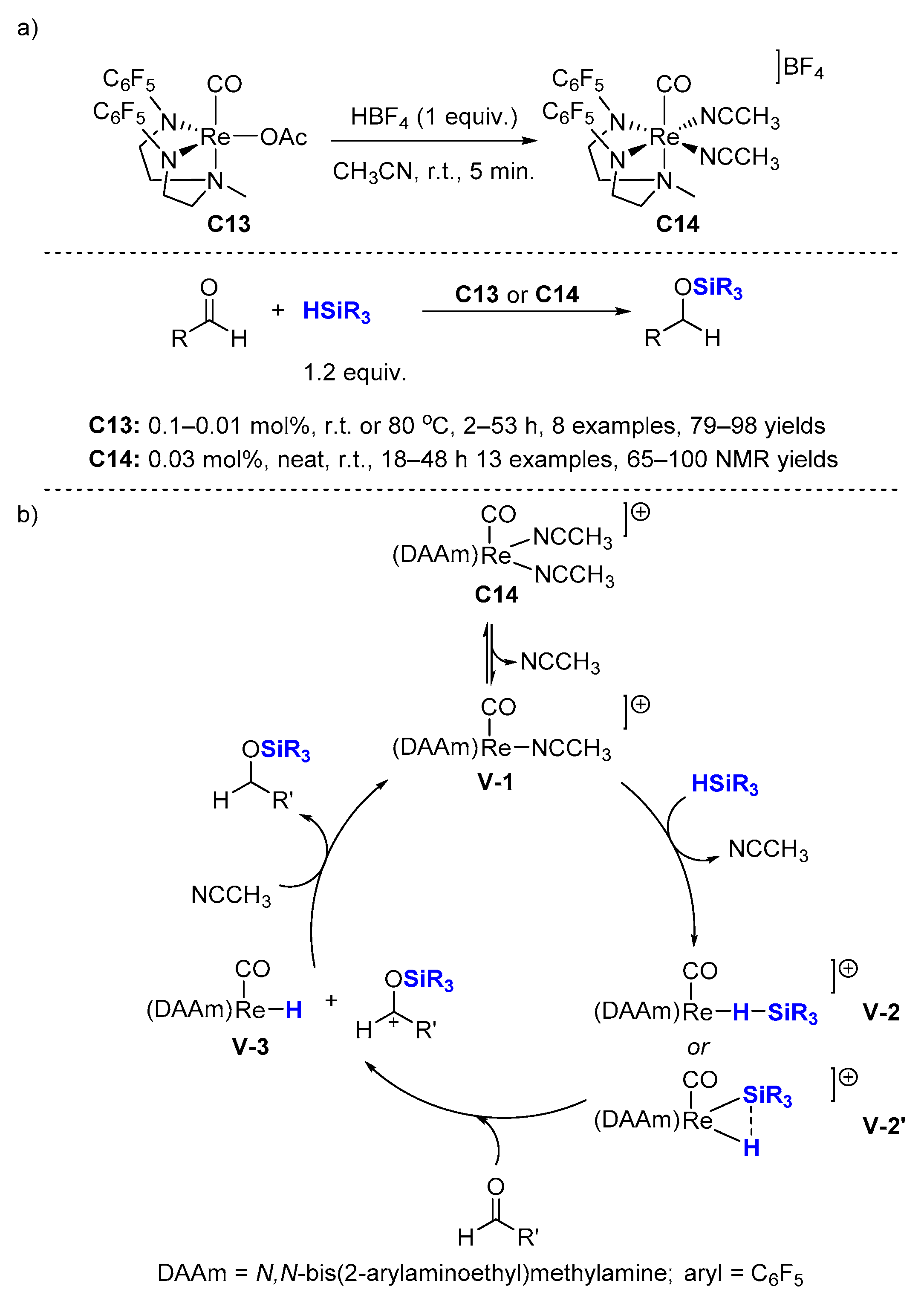
Careful investigation of the hydrosilylation of olefins at poly(ethylene glycol) chain ends and development of a new silyl hydride to avoid side reactions - Shin - 2018 - Journal of Polymer Science

Mechanistic insights into the hydrosilylation of allyl compounds – Evidence for different coexisting reaction pathways - ScienceDirect

Figure 2. Summary of the Hydrosilylation Reaction Mechanism : Characterization and Synthesis of Functionalized Polysilalkylene Siloxane Monomers by Hydrosilylation Reaction : Science and Education Publishing
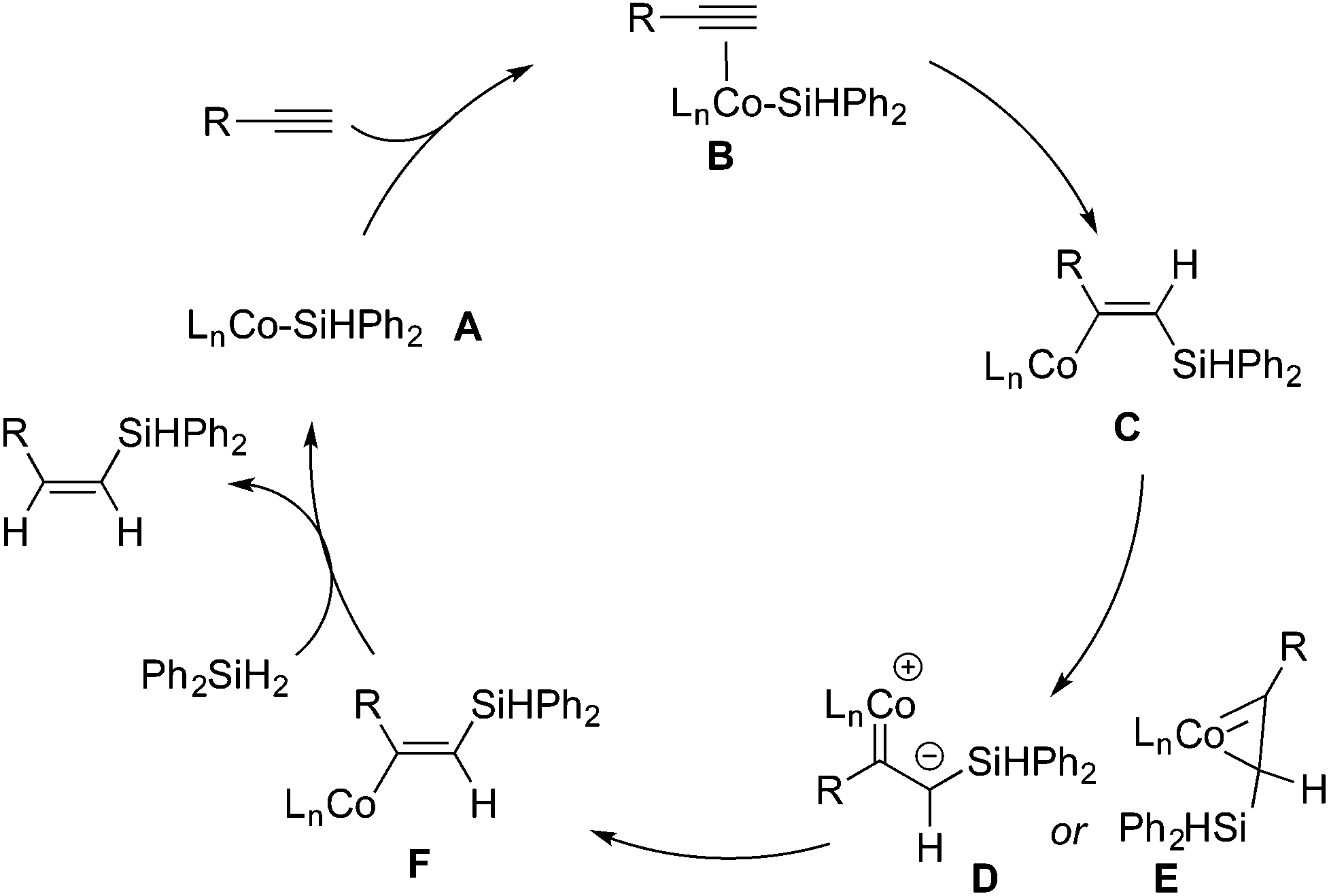
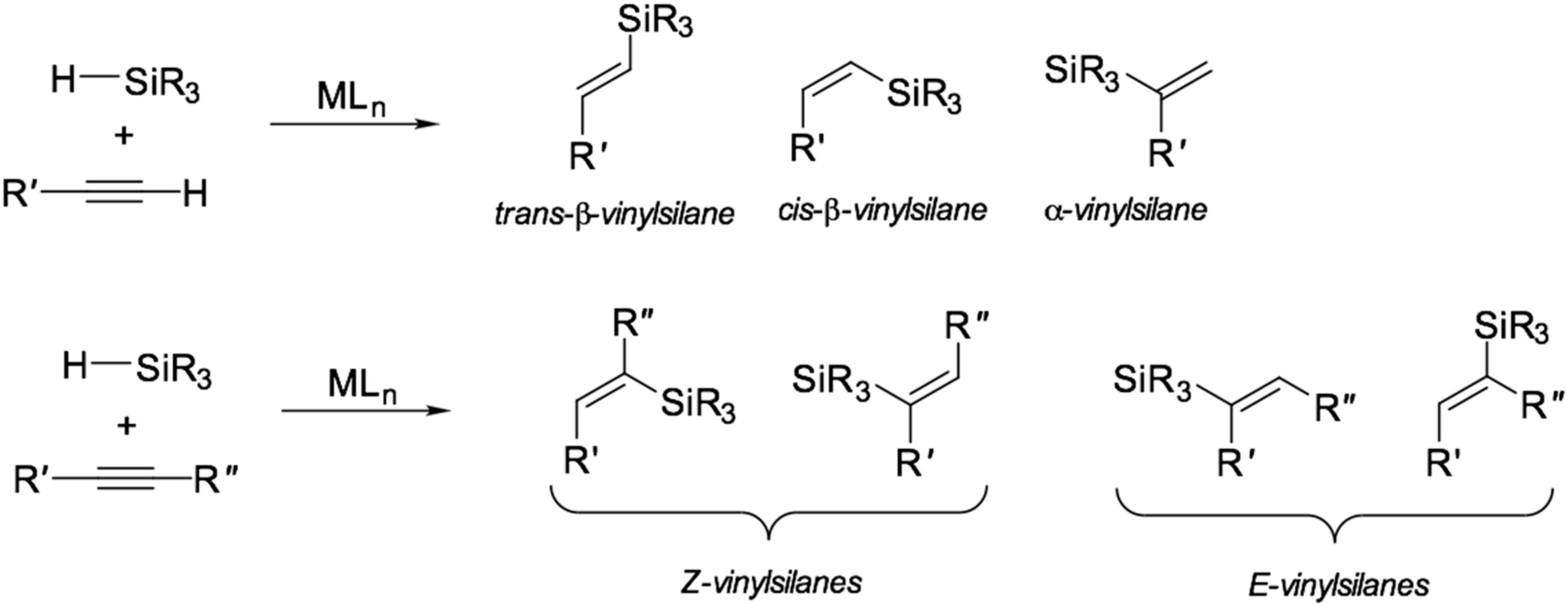
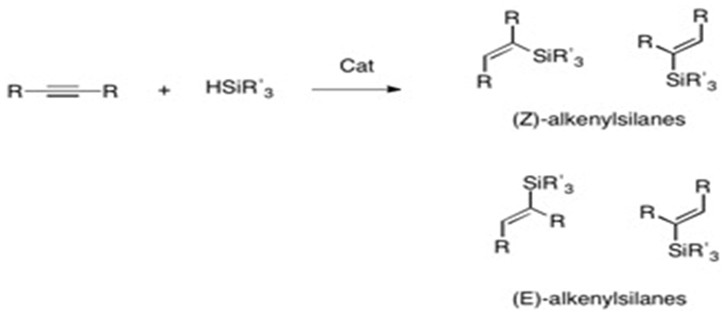
Pincer cobalt complex-catalyzed Z -selective hydrosilylation of terminal alkynes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C7QO00250E
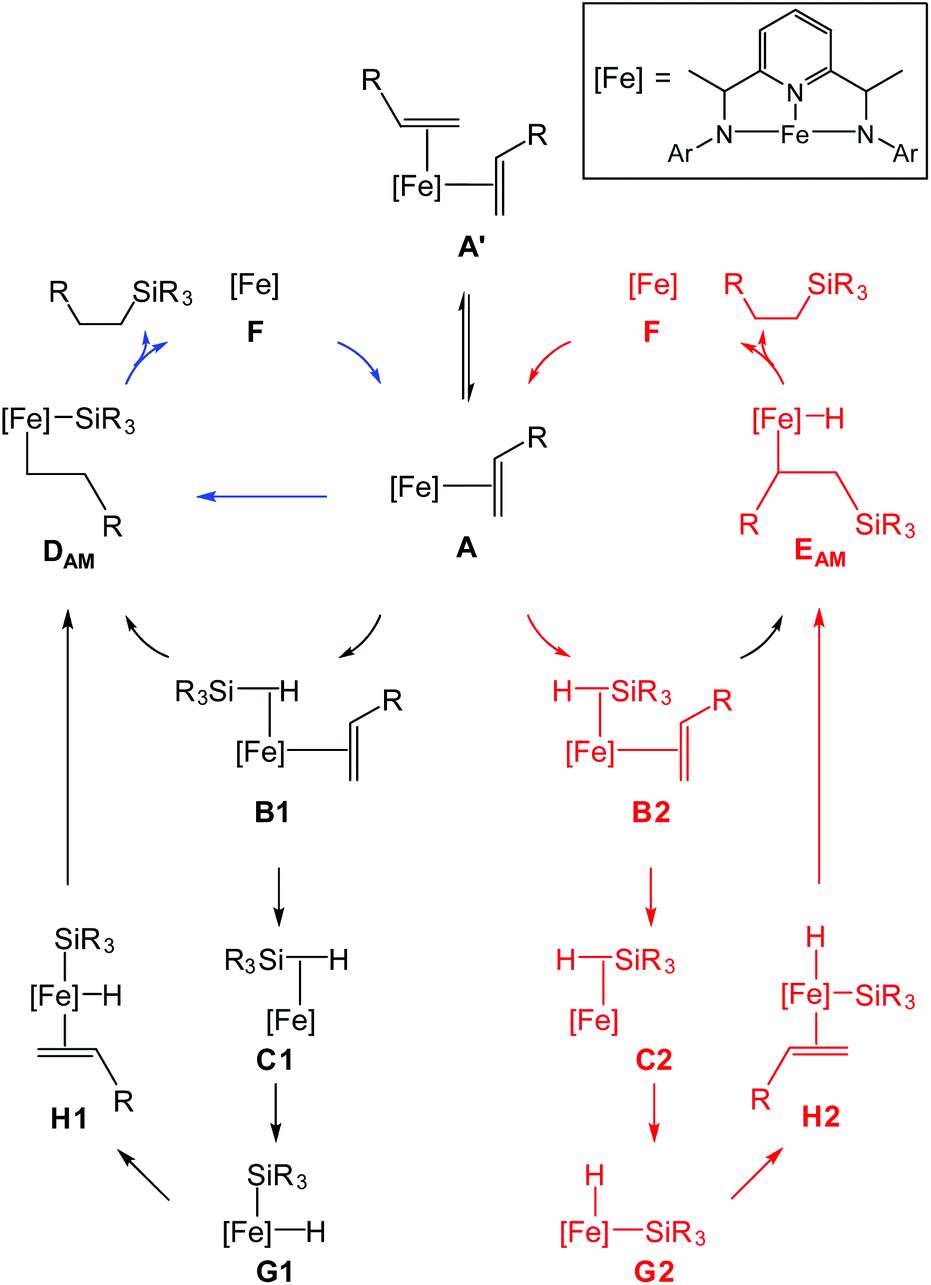
The mechanism for catalytic hydrosilylation by bis(imino)pyridine iron olefin complexes supported by broken symmetry density functional theory - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT02300F
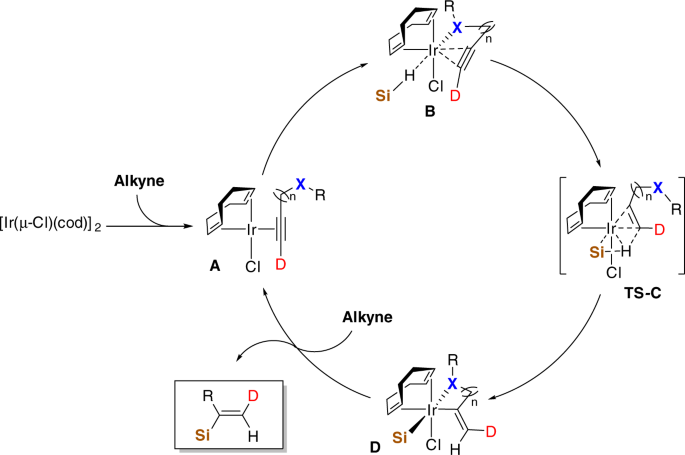
Iridium-catalyzed Markovnikov hydrosilylation of terminal alkynes achieved by using a trimethylsilyl-protected trihydroxysilane | Communications Chemistry
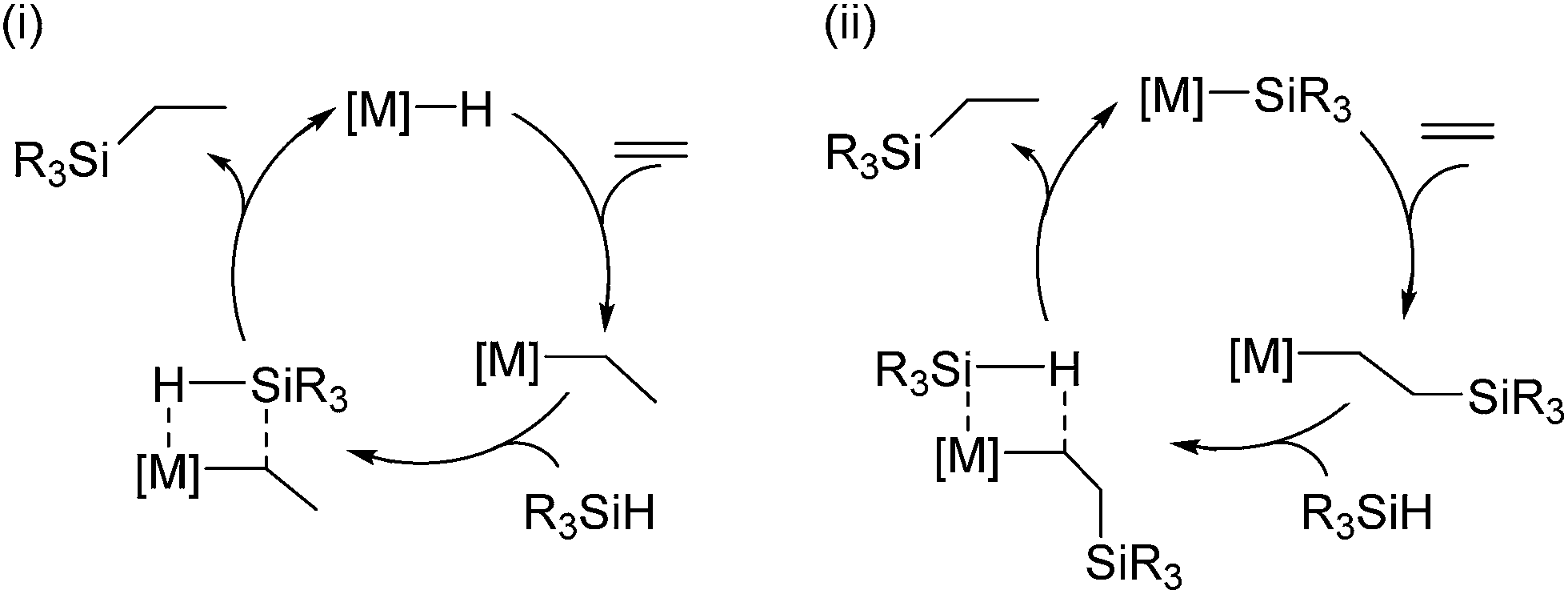
Hydrosilylation reaction of olefins: recent advances and perspectives - RSC Advances (RSC Publishing) DOI:10.1039/C4RA17281G

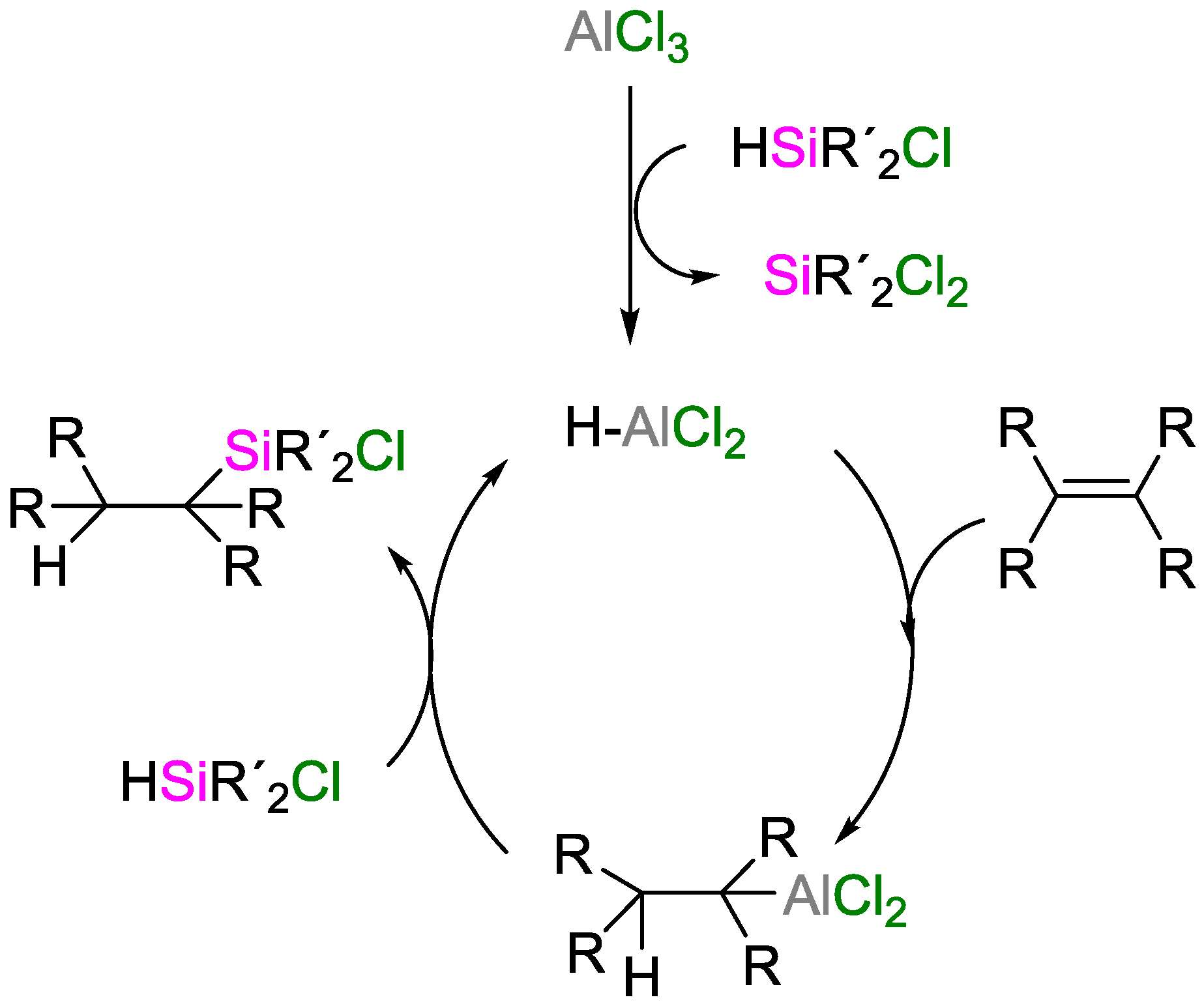
Hydrosilylation of Alkenes with Early Main‐Group Metal Catalysts - Buch - 2006 - Angewandte Chemie International Edition - Wiley Online Library

Importance of the Pd and Surrounding Sites in Hydrosilylation of Internal Alkynes by Palladium–Gold Alloy Catalyst - ScienceDirect

Alkene Hydrosilylation on Oxide‐Supported Pt‐Ligand Single‐Site Catalysts - Chen - 2019 - ChemCatChem - Wiley Online Library
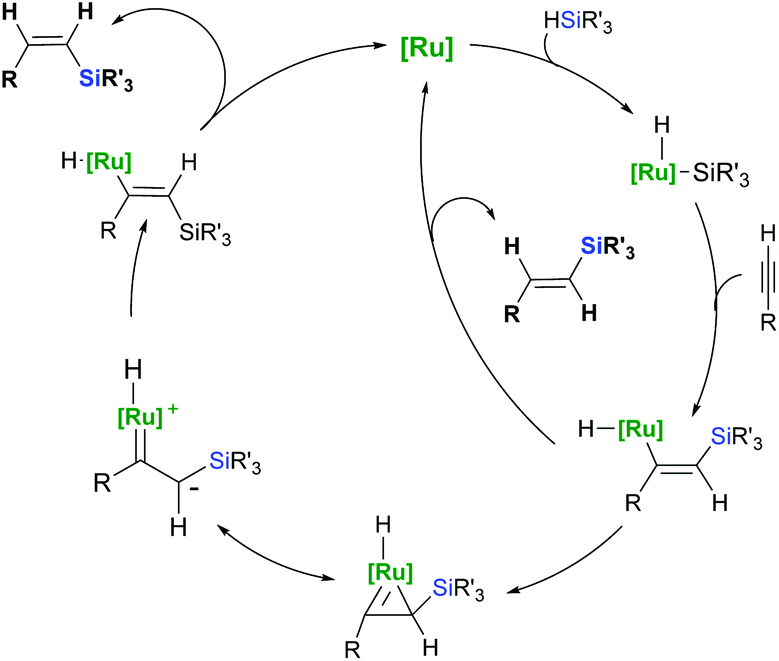
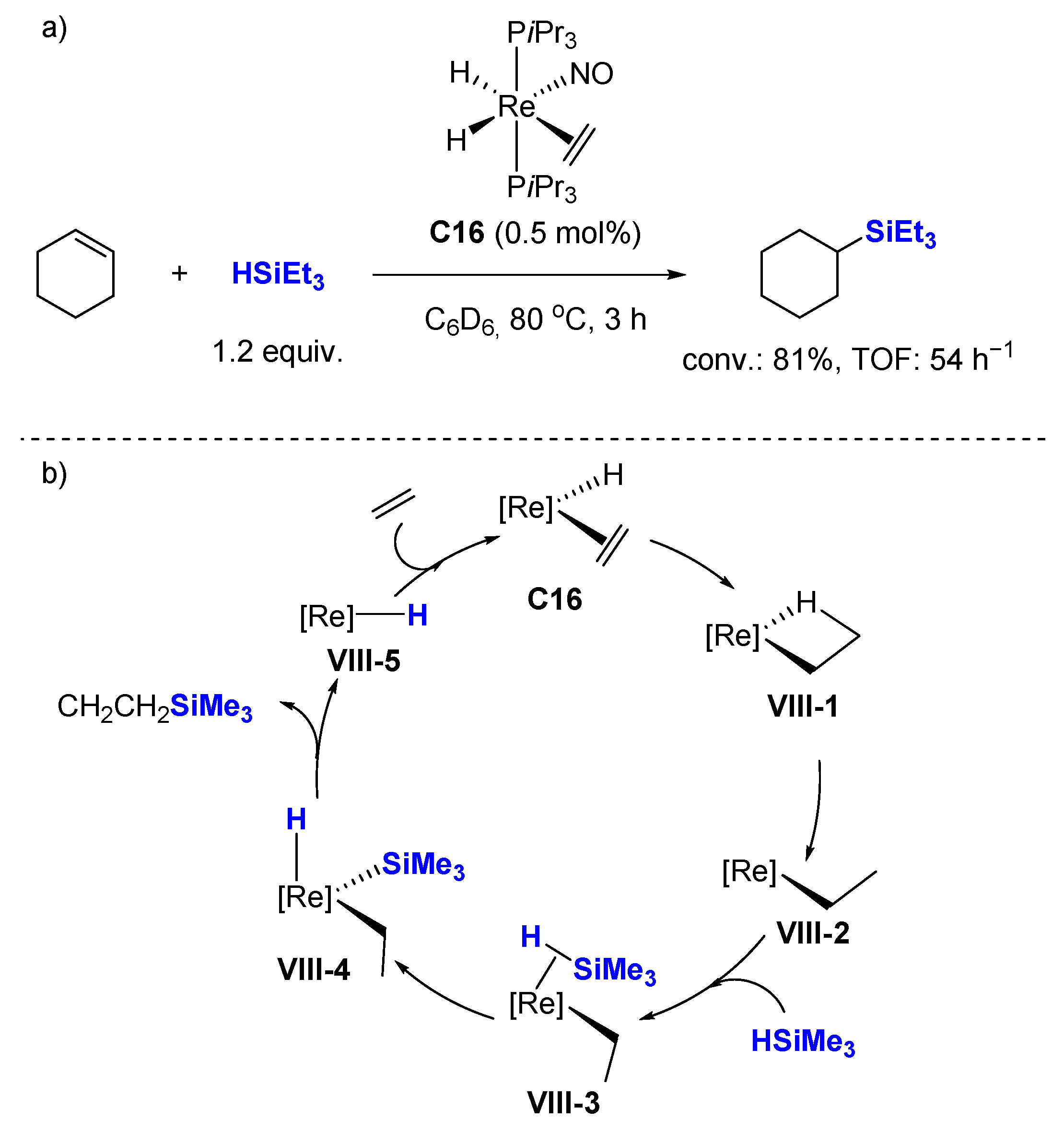
Ruthenium-catalysed hydrosilylation of carbon–carbon multiple bonds - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C6QO00261G
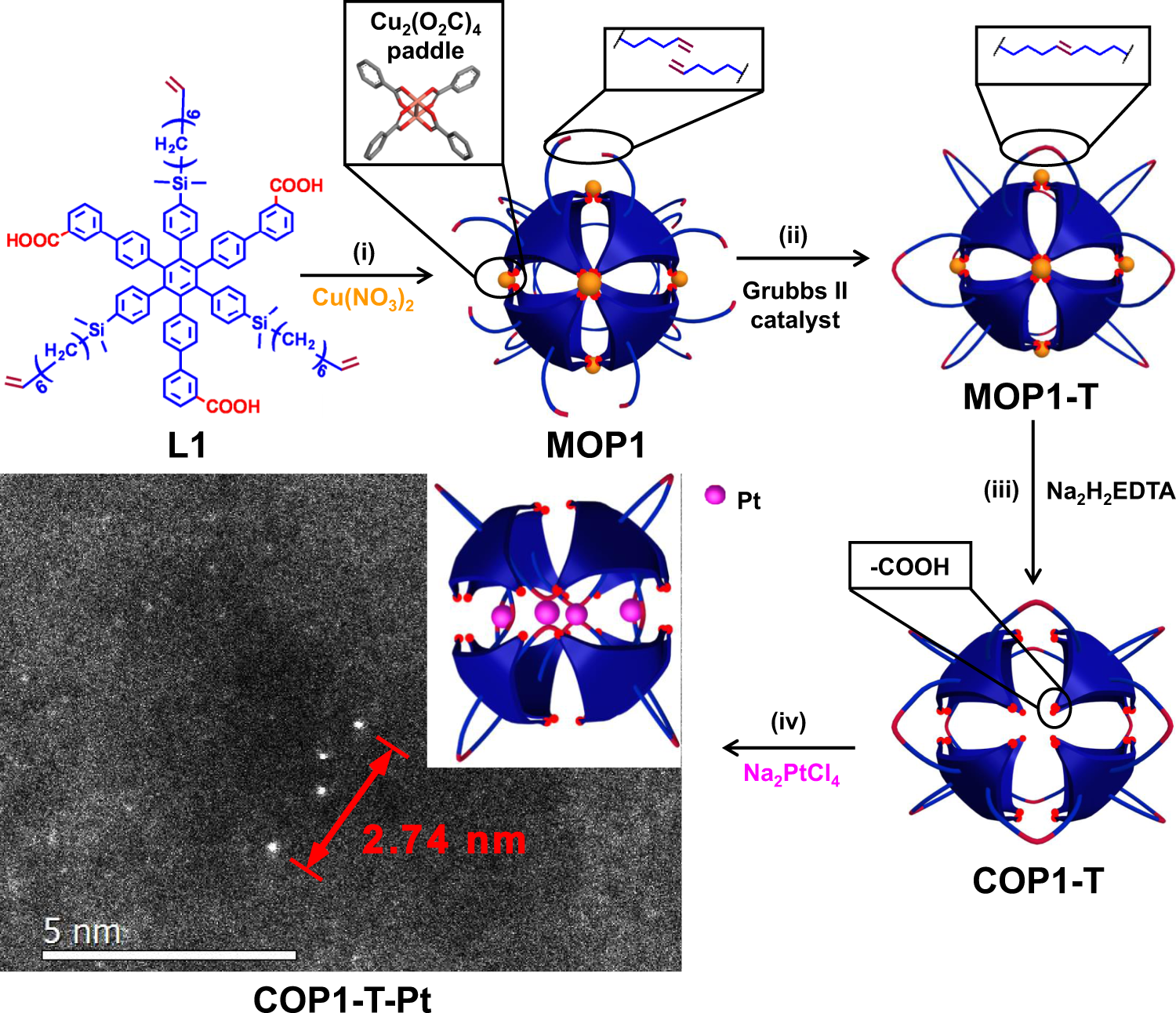
Biomimetic caged platinum catalyst for hydrosilylation reaction with high site selectivity | Nature Communications
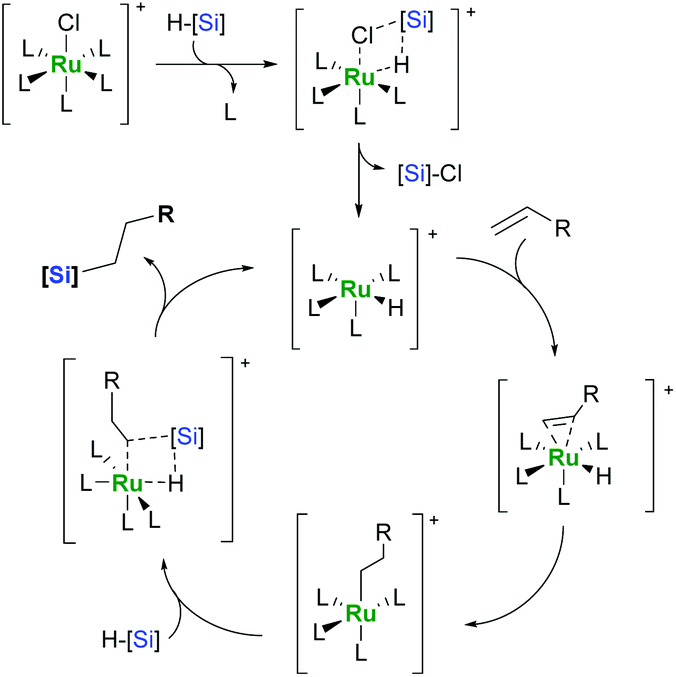
Ruthenium-catalysed hydrosilylation of carbon–carbon multiple bonds - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C6QO00261G