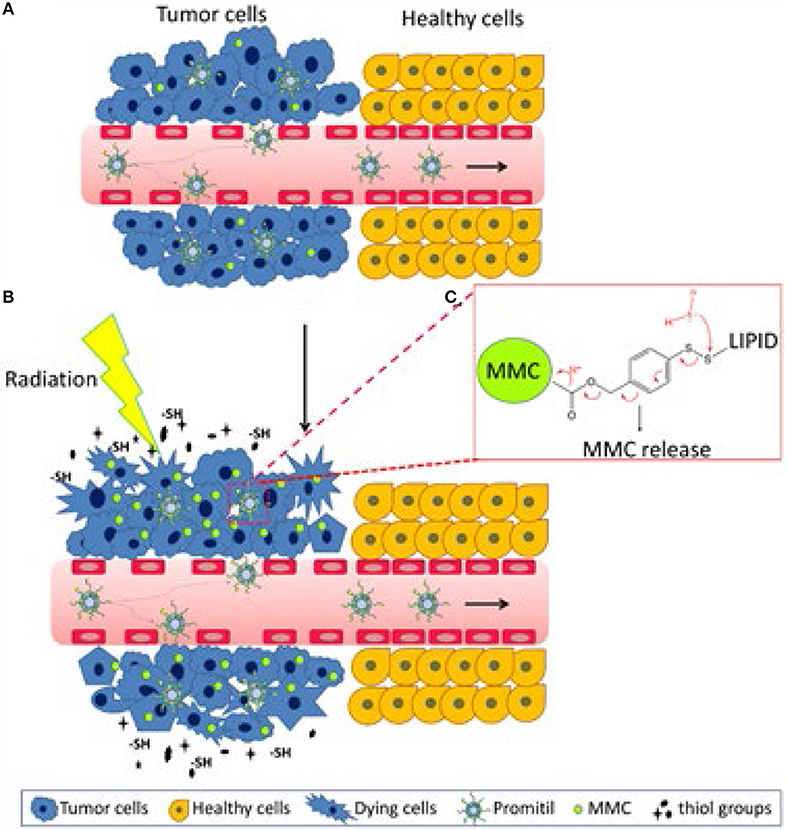
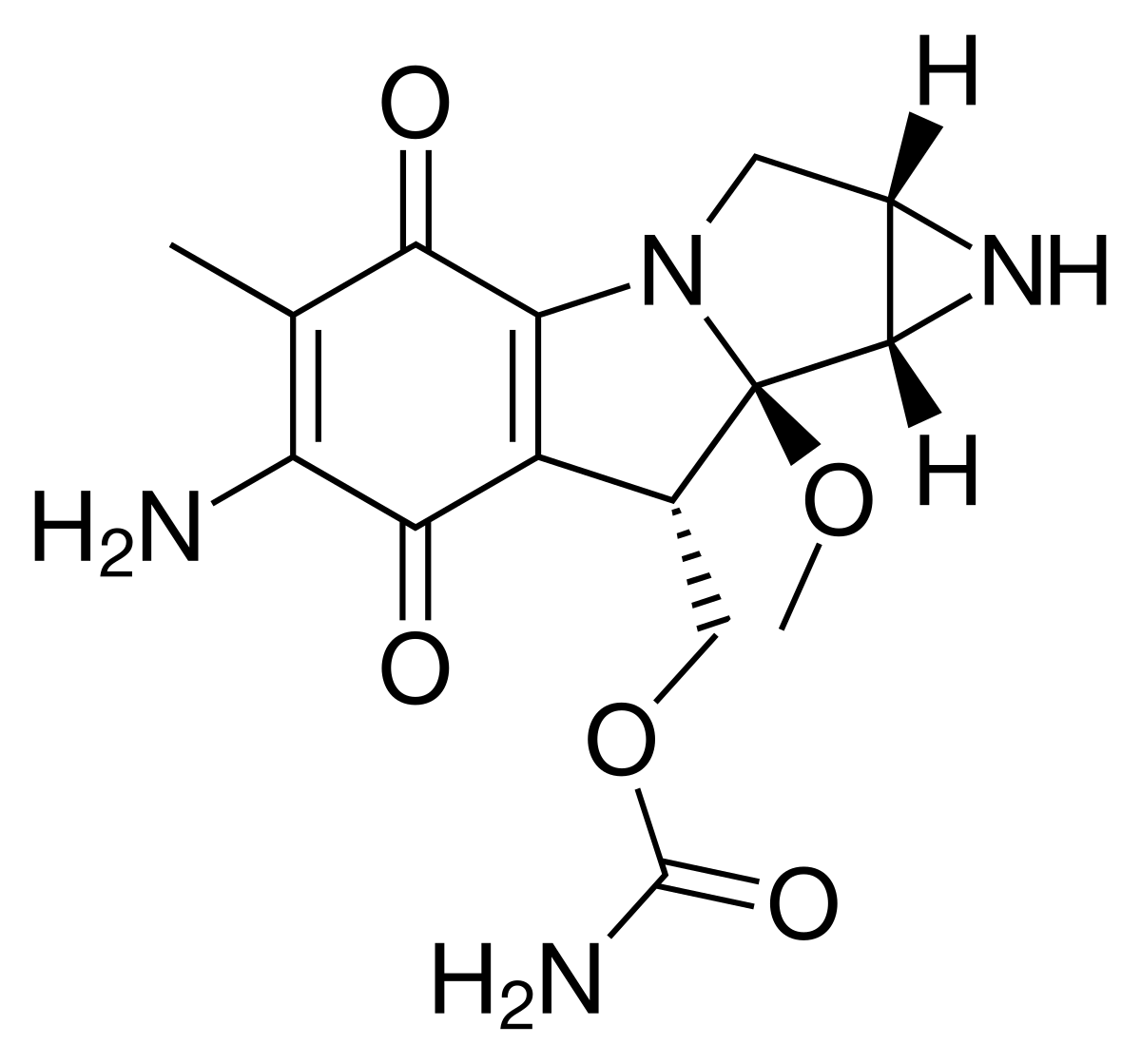
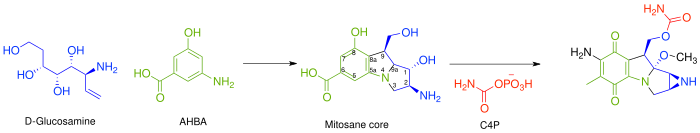
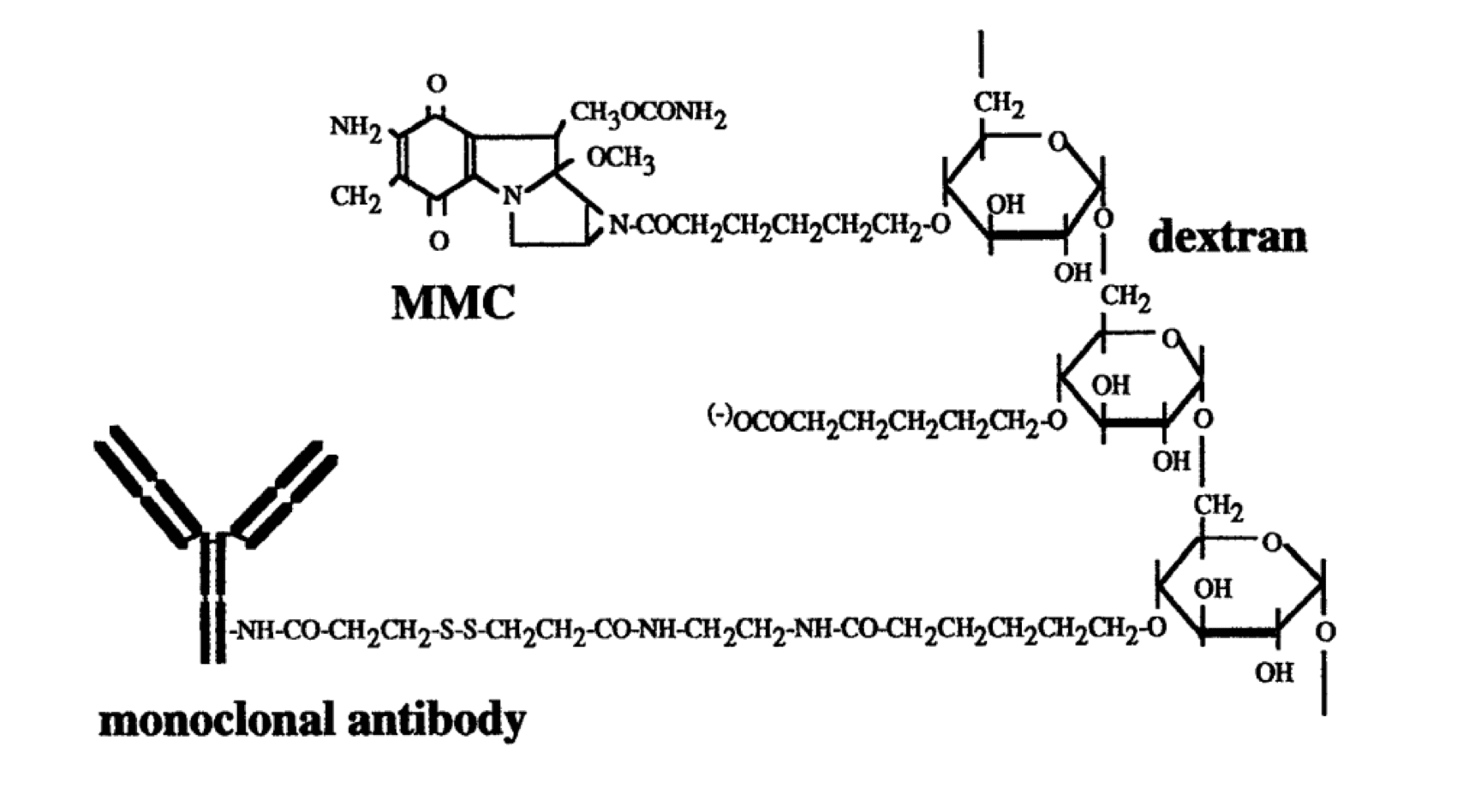
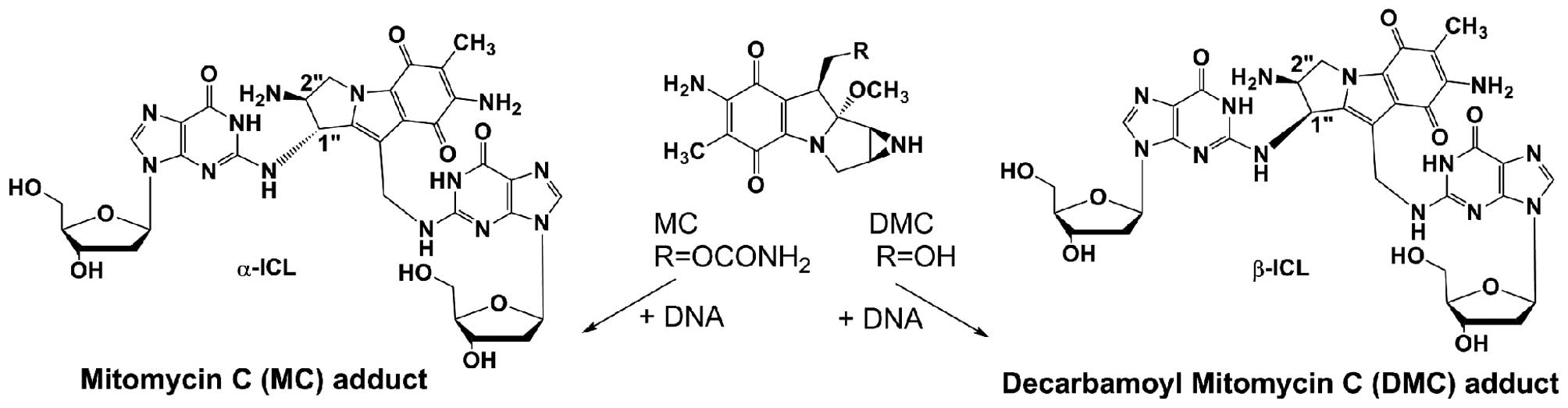
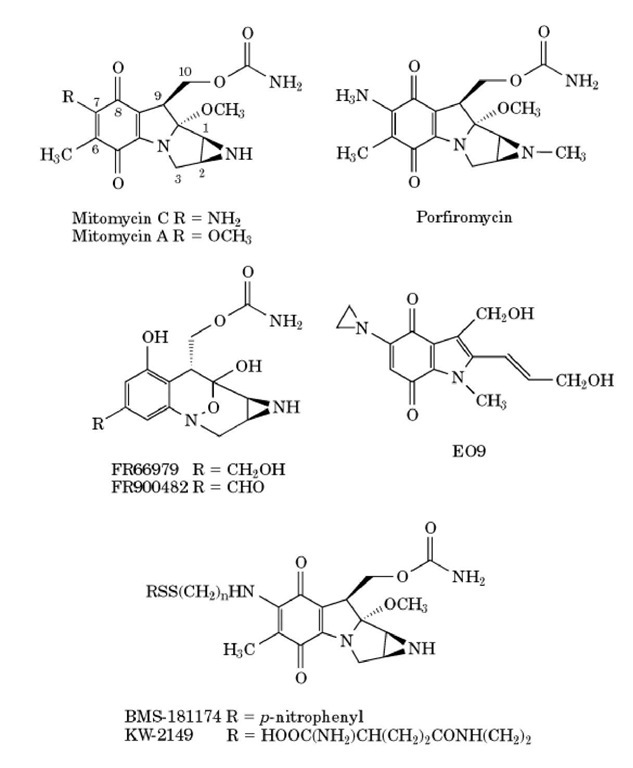
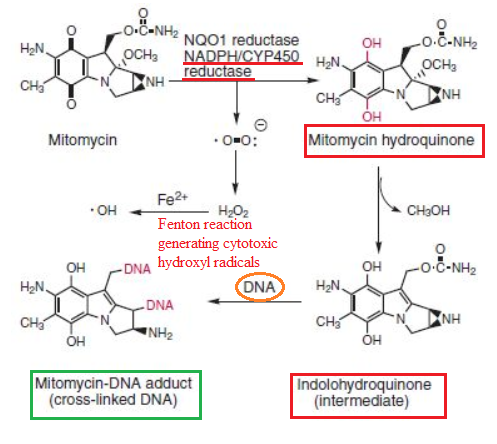
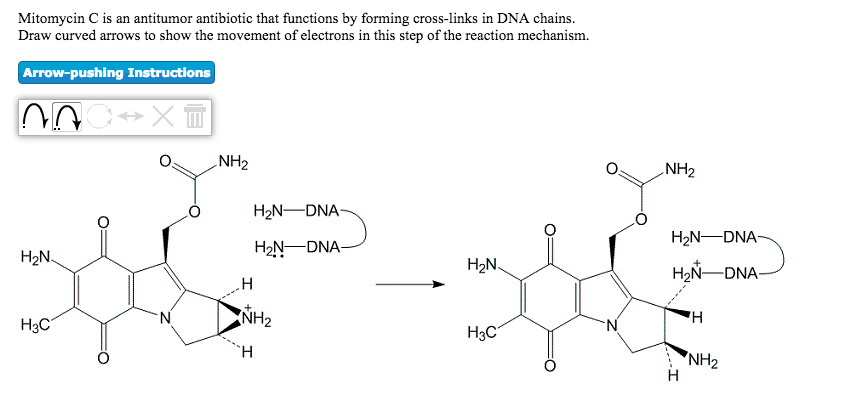
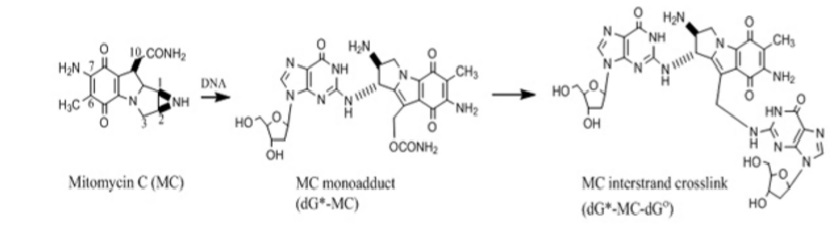
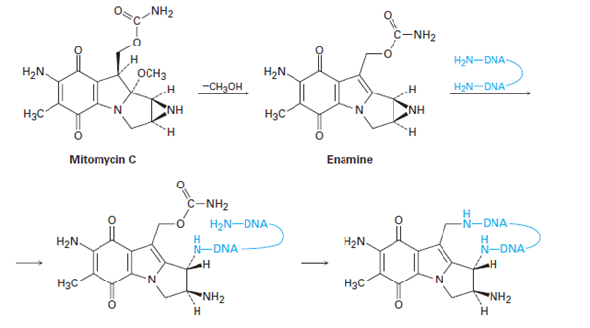
Pegylated liposomal mitomycin C prodrug enhances tolerance of mitomycin C: a phase 1 study in advanced solid tumor patients - Golan - 2015 - Cancer Medicine - Wiley Online Library
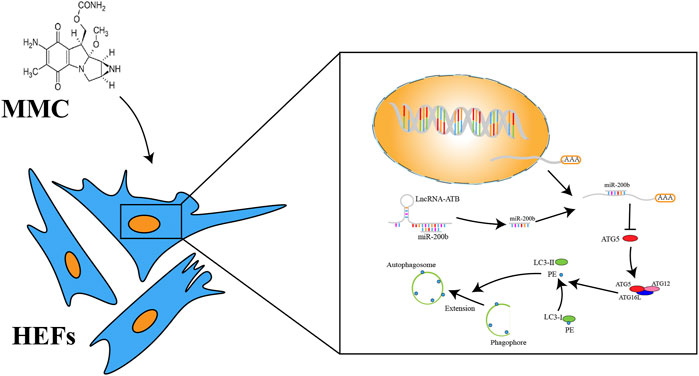
Frontiers | Mitomycin C Inhibits Esophageal Fibrosis by Regulating Cell Apoptosis and Autophagy via lncRNA-ATB and miR-200b | Molecular Biosciences

Characterization of a quinone reductase activity for the mitomycin C binding protein (MRD): Functional switching from a drug-activating enzyme to a drug-binding protein | PNAS

Pharmacologic Studies of a Prodrug of Mitomycin C in Pegylated Liposomes (Promitil®): High Stability in Plasma and Rapid Thiolytic Prodrug Activation in Tissues | Semantic Scholar

LHRH Targeted Chonderosomes of Mitomycin C in Breast Cancer: An In Vitro/ In Vivo Study | Bentham Science
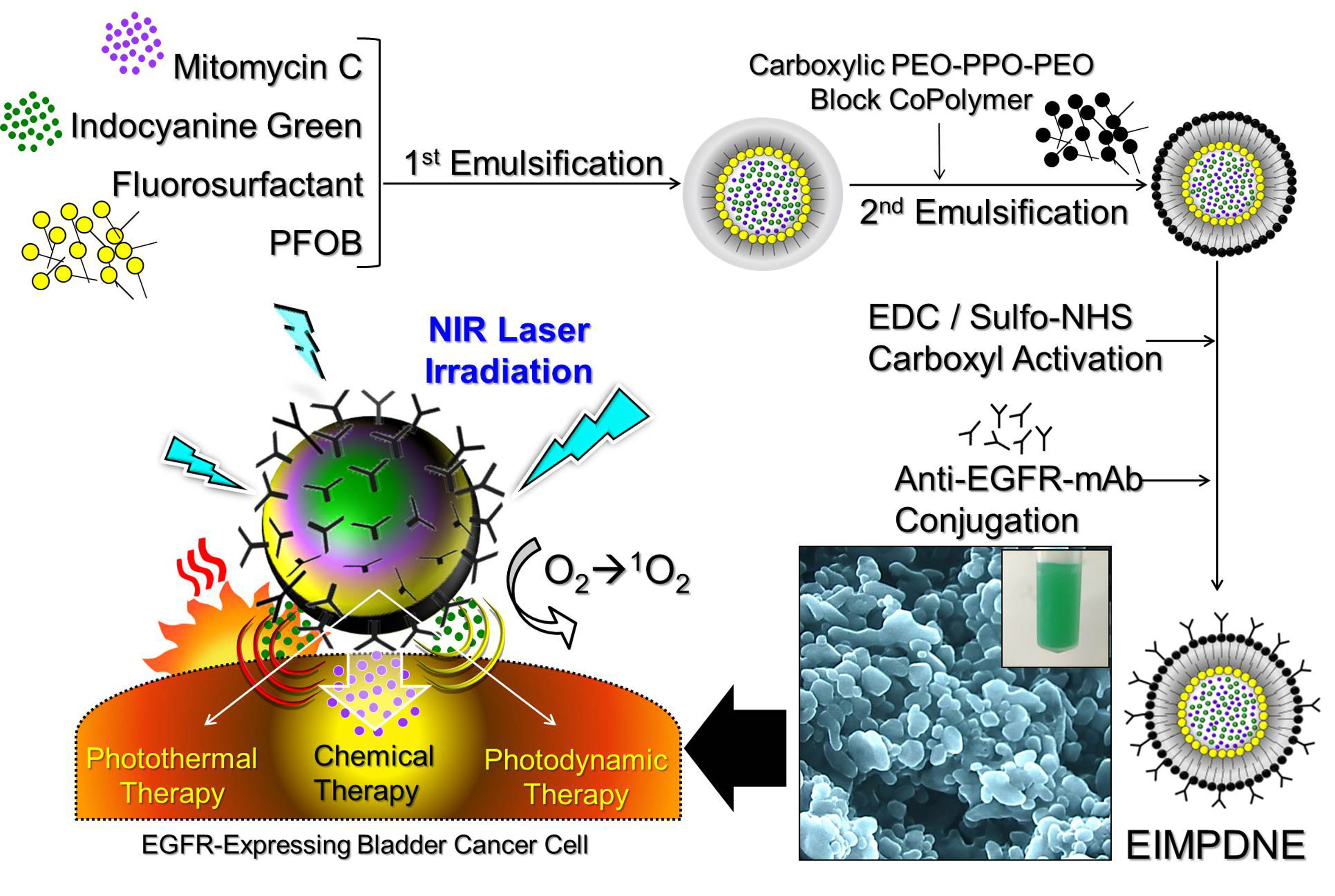
Nanomaterials | Free Full-Text | Anti-EGFR Indocyanine Green-Mitomycin C-Loaded Perfluorocarbon Double Nanoemulsion: A Novel Nanostructure for Targeted Photochemotherapy of Bladder Cancer Cells | HTML

A New Mechanism of Action for the Anticancer Drug Mitomycin C: Mechanism-Based Inhibition of Thioredoxin Reductase | Chemical Research in Toxicology